Kybella
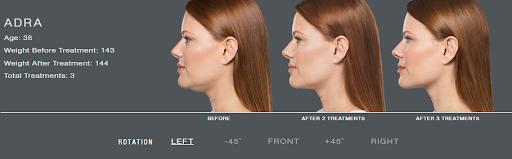
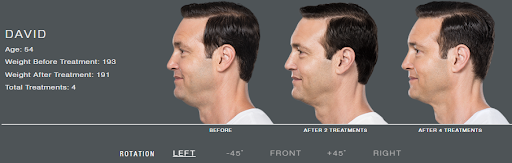
Kybella, or deoxycholic acid, is a naturally occurring molecule which is used to dissolve the fat below the chin. It usually takes a few treatments to get to your goal, however you will see improvement with each treatment. Treatments are spaced a minimum of one month apart.
Frequently Asked Questions:
How many sessions will I need?
It usually takes 2-4 sessions to achieve your goal.
What happens during the treatment?
Injections will only take about 5 minutes. You will feel a slight burning sensation for a few hours after the treatment.
What can I expect after?
You can expect swelling and bruising after injections, however it will lessen with each session. Icing will help decrease these symptoms. This usually last 3-5 days.
What is the after care / Is there any downtime?
- DOWNTIME: Bruising and swelling for about a week
- Drink plenty of fluids to stay hydrated
Treatment
One Session
$800.00
Plan a Visit
Upon booking your appointment a credit/debit card must be on file to reserve your appointment. The card on file will be charged $100 for no shows or same day cancellations.

Contact Us
Schedule at the click of a button and join our mailing list for special offers and events!
Sign up for weekly goodies or schedule your appointment today!
(856)-809-0909
2301 East Evesham Road Voorhees, NJ 08043
Suite 305
info@anumedicalspa.com
Spa Hours
Sunday: Closed
Monday: 9:00 a.m. – 5:00 p.m.
Tuesday: 9:30 a.m. – 7:00 p.m.
Wednesday: 9:00 a.m. – 5:00 p.m.
Thursday: 9:00 a.m. – 5:00 p.m.
Friday: 9:00 a.m. – 5:00 p.m.
Every Other Saturday: 9:00 a.m. – 3:00 p.m.



